Past Issues
Clinical Perspective: Greater Magnitude of Reactive Nodal Uptake Following COVID-19 mRNA Vaccination Compared to Traditional Influenza Vaccination
Yingbing Wang MD*, David Z Chow MD, Shalini Santoshi Indrakanti, James A Scott MD
Massachusetts General Hospital, Department of Radiology, Boston, MA, USA
*Corresponding Author: Yingbing Wang, MD, Massachusetts General Hospital, Department of Radiology, 55 Fruit Street, Wht 4-427, Boston, MA 02114, Tel: 617-726-8350; Fax: 617-726-6165; E-mail: [email protected]
Received Date: July 09, 2021
Publication Date: August 25, 2021
ABSTRACT
Background: Increased axillary nodal uptake ipsilateral to site of COVID-19 vaccination on FDG PET is described in the literature.
Purpose: We hypothesize that novel mRNA COVID-19 vaccines induce greater reactive nodal uptake than traditional Influenza vaccines.
Materials and Methods: We retrospectively reviewed a consecutive series of FDG PET scans performed in patients with recent (<30 days) mRNA-type COVID-19 vaccination during the early vaccination rollout period. The EHR was queried for subset of patients who also had prior FDG PET scans performed within 30 days of intramuscular Influenza vaccination. All relevant FDG PET imaging was reviewed for magnitude of nodal uptake (SUVmax) and assigned a qualitative score of 1, if less than or equal to mediastinum reference, 2 if less than or equal to liver reference, and 3 if greater than liver reference.
Results: During the study period spanning February 22-March 11, 2021, we identified 176 FDG PET scans performed in patients with recent mRNA-type COVID-19 vaccination. 23% of scans showed intense (greater than liver) nodal uptake ipsilateral to the site of injection. All correlative lymph nodes on diagnostic CT remained sub centimeter. 19 patients were identified to have also had prior FDG PET scans performed within 30 days of intramuscular Influenza vaccination; no post-Influenza vaccine FDG PET scans showed any significantly increased axillary nodal uptake.
Conclusion: Our data suggests a significant rate of abnormal ipsilateral nodal uptake following COVID-19 mRNA vaccination and that this rate is likely significantly higher compared to traditional intramuscular Influenza vaccination, possibly due to the highly immunostimulatory effects of the mRNA vaccines.
KEYWORDS: COVID-19; FDG PET; mRNA; Axillary lymph nodes
INTRODUCTION
In keeping with a national, population-wide vaccination campaign to curb SARS-CoV-2 viral transmission as well as morbidity and mortality associated with COVID-19 infection, our institution commenced vaccinations for patients in late January 2021. In rapid succession, three COVID-19 vaccines have been FDA approved to date: Pfizer in November 2020; Moderna in December 2020; and Johnson and Johnson at the end of February 2021. Since the start of the COVID-19 pandemic, multiple articles have chronicled the wide-ranging radiologic manifestations and evolution of active COVID-19 infection [1,2] COVID-19 sequelae [3,4], and more recently, vaccination related findings [5]. In particular, McIntosh LJ, et al. and Mehta N, et al. have described asymmetric F18 Fluorodeoxyglucose avid axillary adenopathy related to COVID vaccination on PET [6,7]. These reports are consistent with our own institutional observations of abnormally increased, reaching intense, axillary nodal uptake, sometimes also associated ipsilateral deltoid muscle uptake, following COVID-19 vaccination. While reactive axillary nodal uptake is a common finding on FDG PET with multifactorial etiologies [8], both the magnitude and the frequency of nodal uptake in the setting of COVID-19 vaccination is notable. Any upper extremity procedure has the potential to induce ipsilateral nodal enlargement and/or increased uptake, and vaccine related nodal uptake in the pre-COVID era has also been reported [9-11]. However, preventative immunization, including the most commonly performed influenza vaccine, pre-date the advent of commercial PET, and yet there are not noticeably higher rates of intense axillary nodal uptake during the seasonal flu vaccination distribution period. As such, we hypothesize that the COVID-19 mRNA vaccinations are associated with greater reactive nodal uptake than the traditional inactivated influenza vaccine. To test our hypothesize, we reviewed a retrospective cohort of patients with FDG PET performed following COVID-19 vaccination to assess overall rate of intense axillary nodal uptake associated with COVID-19 vaccination. We identified a subset of patients who also had prior FDG PET imaging performed within close temporal proximity (<30 days) to Influenza vaccination to allow for comparison of the degree of induced axillary nodal uptake between vaccine types.
MATERIALS AND METHODS
Institutional review board approval was obtained. Consent was waived.
Vaccination history
All FDG PET exams performed between February 22, 2021 and March 11, 2021 on patients with recent (<30 days) COVID-19 exams was included in this retrospective study. The start date marked the day COVID-19 vaccination screening was implemented at clinical FDG PET appointments. The end date was selected so that the COVID-19 vaccination type was limited to one of the two mRNA vaccinations in distribution at the time, i.e. to exclude any patients who could potentially have received the single dose, Johnson and Johnson human adenovirus vector vaccine. Our COVID-19 vaccine screening questionnaire asks for date of most recent COVID-19 vaccine and site of injection. The dose sequence and brand of vaccination was obtained from the electronic health records, if available. The master list of all patients with recent COVID-19 vaccination was used to generate a subsidiary list of patients with any older FDG PET scan performed within 30 days following a documented intramuscular administration of influenza vaccine. All influenza vaccines were inactivated quadrivalent (Sanofi, Cambridge Massachusetts).
FDG PET scans performed in patients recent Johnson and Johnson brand COVID-19 vaccination after February 27, 2021 were excluded.
Image analysis
FDG PET scans performed within 30 days of COVID-19 vaccination were reviewed to measure and score the degree of highest axillary nodal uptake ipsilateral to the site of intramuscular injection. If the whole-body images suggested confounding uptake related to the clinical indication on the vaccinated side (e.g. left breast cancer and left upper extremity vaccination or left upper extremity melanoma and left upper extremity vaccination), the scan (and subject) was excluded.
In subset of patients who also had older FDG PET imaging performed within 30 days of Influenza vaccination, the older FDG PET scan was also reviewed for axillary nodal uptake.
Axillary nodal uptake was measured (SUVmax) and scored as follows: 1=uptake less than mediastinum, including absent uptake; 2= uptake equal or greater than mediastinum but less than liver; 3= uptake equal to or greater than liver.
The largest ipsilateral lymph node was also measured on CT (diagnostic CT when available; if not, CT attenuation correction was used) and scored as follows: 1=short-axis less than or equal to 1 cm and 2=short axis greater than 1 cm.
Clinical follow up
The electronic health records were queried for reports of symptoms prompting COVID-19 PCR testing following vaccination.
FDG PET technique
Each patient fasted for a minimum of 6 hours prior to administration of FDG. A single intravenous injection of FDG was administered if the glucose level was ≤ 250 mg/dL. The patients were scanned approximately 45 minutes after injection in an integrated PET/CT scanner.
Standard clinical whole body FDG PET scanning was performed from skull-base to upper thighs with dose and image acquisition times dependent on the scanner model. For GE Discovery and Siemens Biograph TrueD scanners, the imaging was performed at 3 minutes per bed position for patients with BMI less than 30, 4 minutes per bed position for BMI between 30 and 34, 5 minutes per bed position for BMI between 35 and 44, and 6 minutes per bed position for BMI exceeding 45. Administered FDG dose was 15 mCi for BMI<35, 20 mCi for 35 ≤ BMI < 45, and 25mCi for BMI ≥45. For Siemens mCT Flow scanners, the imaging was performed at the equivalent of 2 minutes per bed position for BMI <25, 3 minutes for 25 ≤ BMI <30, 4 minutes for 30 ≤ BMI <35, 5 minutes for 35 ≤ BMI <45, and 6 minutes for BMI ≥ 45. Administered FDG dose was 10 mCi for BMI < 45 and 15 mCi for BMI ≥ 45. All technical equipment was in compliance with ACR accreditation requirements at time of imaging.
RESULTS
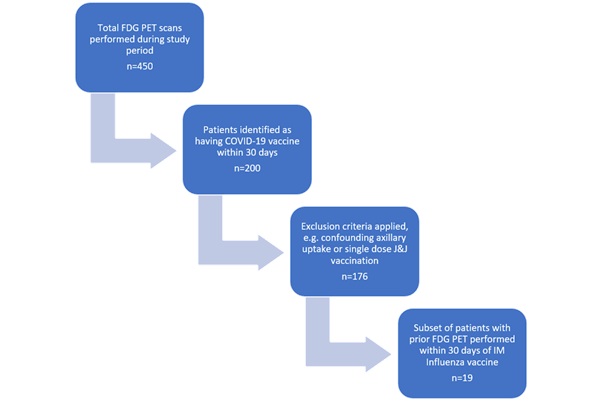
Figure 1 shows the patient selection process and outcomes. A total of 450 clinical FDG PET exams were performed during the study period. The COVID-19 vaccination screening process identified 200 patients who had received a recent COVID-19 vaccine. After excluding for confounding axillary uptake and any non-mRNA vaccines, our cohort included 176 patients. A subset of 19 patients had prior FDG PET scans within 30 days of intramuscular influenza vaccination. The interval between FDG PET scanning and COVID-19 vaccination ranged from 1-21 days; the interval between older FDG PET scanning and Influenza vaccination ranged from 1-29 days.
Figure 1: Patient selection criteria.
Table 1 shows patient demographics and nodal uptake score categorized separately for FDG PET scans performed in (1) all patients with recent COVID-19 vaccination and (2) subset of patients with both recent COVID-19 vaccination and older comparison FDG PET scan performed within 30 days of prior Influenza vaccination.
Table 1: Patient demographics.
|
Recent COVID-19 vaccine (n=176) |
Recent COVID-19 vaccine and prior FDG PET within 30 days of Influenza vaccine (n=19) |
|
|
Sex |
||
|
Males |
88 |
8 |
|
Females |
88 |
11 |
|
Age |
72±6 |
70±10 |
|
PET-Vaccine interval |
||
|
COVID-19 |
12±5 |
12±7 |
|
Influenza |
N/A |
18±11 |
|
Laterality of COVID-19 vaccination |
||
|
Right |
86 |
6 |
|
Left |
90 |
13 |
|
COVID-19 Vaccine type |
||
|
Pfizer |
60 |
5 |
|
Moderna |
40 |
2 |
|
Unknown |
76 |
12 |
|
Dose sequence |
||
|
1st dose |
87 |
8 |
|
2nd dose |
54 |
5 |
|
Unknown |
35 |
6 |
|
Ipsilateral nodal uptake score following COVID-19 vaccination |
||
|
1 |
131 |
12 |
|
2 |
10 |
0 |
|
3 |
35 |
7 |
Figure 2A plots magnitude of uptake nodal uptake against days since vaccination following COVID-19 (blue) and Influenza vaccination (orange). COVID-19 related nodal uptake, measured as SUVmax, ranged from 0 to 6.6. 37% (7/19) scans demonstrated nodal uptake greater than liver (score 3); the remaining scans showed nodal uptake less than or equal to mediastinum. None of the FDG PET exams performed within 30 days of an influenza vaccine showed any uptake greater than mediastinum and only 4/19 (21%) scans showed any appreciable uptake above background.
Figure 2B plots differences in magnitude of nodal uptake between post-COVID vaccine and post-Influenza vaccine scans across patients. Uptake above zero line indicates greater post-COVID vaccine nodal uptake; uptake below zero line indicates greater post-Influenza vaccine nodal uptake.
Figure 2: Patients with both FDG-PET within 30 days of COVID-19 vaccination and older FDG PET performed within 30 days of Influenza vaccination. A: Scatterplot relating axillary nodal SUVmax and days since COVID-19 vaccination (blue) and days since Influenza vaccination (orange). B: Plot relating differences between SUVmax of highest nodal uptake status post COVID-19 vaccine versus Influenza vaccine. Values above zero line indicate higher COVID-19 vaccine related nodal uptake.
Among our cohort, 2 patients with high axillary nodal uptake are documented to have undergone COVID-19 PCR testing within 3 days of their 2nd vaccine dose.
DISCUSSION
The development of mRNA vaccines against COVID-19 is a breakthrough in the national strategy against COVID-19. It has also presented new curiosities and diagnostic challenges when imaging patients who have had recent vaccinations.
Immune responses among individuals are highly variable [12], and we chose to perform our analysis of axillary nodal uptake on patients with both post-COVID vaccine and post-Influenza vaccine FDG PET scans so each subject could serve as his/her own internal control. Overall, 20% of subjects show asymmetric and grade 3 (intense), defined as greater than liver background, axillary nodal uptake following recent COVID vaccination. In the small sub-cohort of patients with both recent FDG PET after COVID-19 vaccination as well as remote FDG PET after Influenza vaccination, 37% of post-COVID-19 vaccine PET scans shows intense axillary nodal uptake while none of the post-Influenza vaccine scans shows significant uptake (Figure 3). Although some correlative lymph nodes on CT measured slightly asymmetrically larger than contralateral nodes, none exceeded 1 cm in short axis diameter.
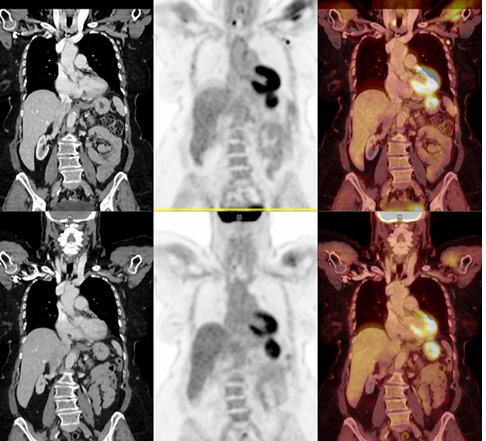
Figure 3: 70 year old female with diffuse large B cell lymphoma of the left lower extremity with COVID-19 vaccination in the left deltoid muscle 21 days prior to her FDG PET scan (top). SUVmax of the highest lymph nodes was 5.6 (short axis 9 mm correlative node on CT). An older FDG PET scan (bottom) was performed 19 days following intramuscular Influenza vaccination. Laterality of Influenza injection is unknown but neither axilla showed any increased uptake.
The mRNA vaccines are designed to be immunostimulatory, and therefore not surprisingly more immunogenic than traditional vaccine types. Lymph nodes are secondary lymphoid organs and increased nodal uptake on FDG PET can be biomarkers of immune system activation. Whether presence of significant nodal uptake is correlated with post-vaccine symptoms is beyond the scope of this study. However, in clinical trials, a large majority (up to 90.1% for Moderna vaccine) of patients experience at least some local symptoms (most commonly injection site pain) following either dose of vaccine [13,14].
We did not find a relationship between sequence of vaccine dosing or manufacturer, however some of this data was unavailable, possibly contributing to an inadequate sample size to observe an effect. It is also interesting that we did not show a significant relationship between interval since COVID-19 vaccination and magnitude of uptake. It is axiomatic that reactive uptake subsides over time, and were multi-time point imaging available, we likely would have observed higher uptake within the same subject at timepoints nearer to the date of injection and less uptake further from the date of injection. This in-patient trend is likely obscured by high levels of variability across patients and the relatively small sample size.
Limitations to our study include a small sample size of patients with both recent FDG PET imaging in close temporal proximity to COVID-19 and available prior FDG PET in close temporal proximity to Influenza vaccination, however maintaining each patient as his/her own internal control was an important study design consideration. We were also only able to provide limited clinical correlation between nodal uptake and either local or systemic symptoms, as detailed post-vaccine symptom data, while collected by our institution, is not available through the electronic health record system. However, any reports of fever or severe symptoms does trigger contact by health services and the patient is urged to undergo COVID-19 PCR testing. Among our cohort, 2 patients with high axillary nodal uptake are documented to have undergone COVID-19 PCR testing within 3 days of their 2nd vaccine dose; the results of both tests were negative.
In conclusion, our data collected during the early vaccine rollout period revealed a high rate of reactive nodal uptake following recent COVID-19 vaccination and both the rate and magnitude of uptake are likely much higher than commonly observed in a similar time interval following Influenza vaccination. The implications are that post-vaccine axillary nodal uptake can confound staging in patients with primary malignancies that have a predilection for axillary or sub pectoral nodal spread. It follows that a careful vaccine history, potentially taken at the beginning of a PET imaging appointment, can aid the image interpretation process, particularly if the patient received the vaccine a local pharmacy, e.g. CVS, and vaccine documentation may not be part of the EHR.
CONFLICTS OF INTEREST
The authors have no conflicts of interest or financial support to disclose.
CONSENT
The study was approved by the authors’ institutional review board. Informed consent was waived.
REFERENCES
- Joob B, Wiwanitkit V. (2020). 18F-FDG PET/CT and COVID-19. Eur J Nucl Med Mol Imaging. 47:1348.
- Delorme C, Paccoud O, Kas A, Hesters A, Bombois S, et al. (2020). COVID-19-related encephalopathy: a case series with brain FDG-positron-emission tomography/computed tomography findings. Eur J Neurol. 27(12):2651-2657.
- Liu M, Lv F, Huang Y, Xiao K. (2021). Follow-Up Study of the Chest CT Characteristics of COVID-19 Survivors Seven Months After Recovery. Front Med (Lausanne). Online ahead of print. 8:636298. doi: 10.3389/fmed.2021.636298.
- Sollini M, Ciccarelli M, Cecconi M, Aghemo A, Morelli P, et al. (2020). Vasculitis changes in COVID-19 survivors with persistent symptoms: an [(18)F]FDG-PET/CT study. Eur J Nucl Med Mol Imaging. 48(5):1460-1466.
- Doss M, Nakhoda SK, Li Y, Yu JQ. (2021). COVID-19 Vaccine-Related Local FDG Uptake. Clin Nucl Med. 46 (5):439-441.
- McIntosh LJ, Bankier AA, Vijayaraghavan GR, Licho R, Rosen MP. (2021). COVID-19 Vaccination-Related Uptake on FDG PET/CT: An Emerging Dilemma and Suggestions for Management. AJR Am J Roentgenol. Online ahead of print. doi: 10.2214/AJR.21.25728.
- Mehta N, Sales RM, Babagbemi K, Levy AD, McGrath AL, et al. (2021). Unilateral axillary Adenopathy in the setting of COVID-19 vaccine. Clin Imaging. 75:12-15. doi: 10.1016/j.clinimag.2021.01.016.
- Hofman MS, Hicks RJ. (2016). How We Read Oncologic FDG PET/CT. Cancer Imaging. 16:35.
- Shirone N, Shinkai T, Yamane T, Uto F, Yoshimura H, et al. (2012). Axillary lymph node accumulation on FDG-PET/CT after influenza vaccination. Ann Nucl Med. 26(3):248-252.
- Thomassen A, Lerberg Nielsen A, Gerke O, Johansen A, Petersen H. (2011). Duration of 18F-FDG avidity in lymph nodes after pandemic H1N1v and seasonal influenza vaccination. Eur J Nucl Med Mol Imaging. 38(5):894-898.
- Williams G, Joyce RM, Parker JA. (2006). False-positive axillary lymph node on FDG-PET/CT scan resulting from immunization. Clin Nucl Med. 31:731-732.
- Brodin P, Davis MM. (2017). Human immune system variation. Nat Rev Immunol. 17:21-29.
- Baden LR, El Sahly HM, Essink B, Kotloff K, Frey S, et al. (2021). Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N Engl J Med. 384:403-416.
- Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, et al. (2020). Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N Engl J Med. 383:2603-2615.
Citation: Wang Y, et al. (2021). Clinical Perspective: Greater Magnitude of Reactive Nodal Uptake Following COVID-19 mRNA Vaccination Compared to Traditional Influenza Vaccination. Clin Res. 2(1):02.
Copyright: Wang Y, et al. © (2021). This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
 Abstract
Abstract  PDF
PDF